Testing of RAAD
OVERVIEW:
The device will be tested on students by measuring their muscle activity using electromyography ( EMG) without the device and while wearing the device. The muscle potential activity while wearing the device should be significantly less than while not wearing the device. Measurement and analysis of this potential will determine whether the device is working as intended. The device will be working the biceps and triceps although there will be spill-over activity to some other muscles. The arm device should facilitate movement with less arm exertion. This should help the patient maintain physical activity with less fatigue.
The device will be tested on students by measuring their muscle activity using electromyography ( EMG) without the device and while wearing the device. The muscle potential activity while wearing the device should be significantly less than while not wearing the device. Measurement and analysis of this potential will determine whether the device is working as intended. The device will be working the biceps and triceps although there will be spill-over activity to some other muscles. The arm device should facilitate movement with less arm exertion. This should help the patient maintain physical activity with less fatigue.
DETAILED EXPLANATION OF TESTING:
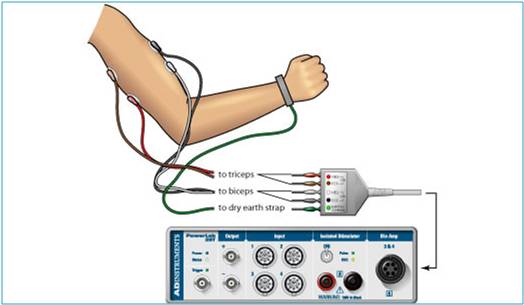
EMG is a technique for evaluating and recording the electrical activity produced by the skeletal muscles. We will use surface electrodes and a computerized data acquisition system to monitor muscle activity (or electromyograms) to test muscle activity when performing the motion without the device and while performing the motion with the device. Surface EMG, a non-invasive test, as opposed to intramuscular EMG using needles and wire, will be used to test the functionality of the device. The adhesive surface electrodes will be placed on the biceps and triceps. The adhesive allows for the easy removal after testing is complete. An electromyogram detects the electrical potential generated by the muscle cells when these cells are neurologically activated. The electrical source is the muscle membrane potential of about -90 mV. EMG potentials range between 50 μV and up to 30 μV. Typical repetition rate of muscle motor firing is about 7-20Hz which depends on the size of the muscle. Muscle tissue at rest is normally electrically inactive. When the muscle is voluntarily contracted, action potentials begin to appear. As the strength of the muscle contraction is increased, more and more muscle fibers produce action potentials. When the muscle is fully contracted, there will be a group of action potentials of varying rates and amplitudes. With the device on, the muscle activity during muscle contraction should be significantly less than without the device.
The patient will be secured into the device by the sensor cuff on the wrist as well as straps around the arm and bicep. The metal frame will support the arm though the patient will not be in contact with the metal. The device will have cushioning material to provide comfort and electrical insulation. Any metal part of the frame that would be in contact with the subject will be insulated.
Each subject will have the same tests performed and it will take about the same amount of time. Test subjects will be studied once during the semester. Testing is not expected to take more than an hour. There is no justified need for follow up trials. Computer software (BIOPAC STUDENT LAB) as well as surface electrodes will be utilized in order to acquire muscle activation data that will be analyzed. This data will consist of an EMG that will be interpreted by the group to determine if the device is working adequately. We plan on testing this device on 20 participants. Each participant will be asked to perform the same motion with and without the device. These will be compared to determine how closely the device approaches physiological range of motion. The study will take place at The College of New Jersey. The investigator s include all team members: Christina Paparella, Andrew Levitsky, Danny Molina, Greg VanNess and Christian Mejia. Christina, Danny and Andrew have performed a similar lab using BIOPAC and specifically testing EMG in the Fall of 2010 in Physiological Systems Laboratory I. The design team has the BIOPAC EMG lesson used in the lab and will follow procedures for interpreting data accordingly.
In order to test the device, BIOPAC data acquisition unit (MP36) will be used to convert the electrical signal of the muscles into interpretable data on a computer. BIOPAC disposable vinyl electrodes (El503) are surface electrodes that are placed on the skin in order to measure motor neuron impulses to be sent to the computer acquisition unit. For optimal electrode adhesion, the electrodes should be placed on the skin at least five minutes before the start of the calibration procedure. Each of the pinch connectors on the end of the electrode cable needs to be attached to a specific electrode. The electrode cables are each a different color to ensure that each cable is connected to the proper electrode. Other important components for testing include the BIOPAC electrode lead set (SS2L), BIOPAC electrode gel (GEL1) and abrasive pad (ELPAD), computer system and BIOPAC Student Lab 3.7.
List of Materials:
BIOPAC data acquisition unit (MP36)
BIOPAC electrode lead set (SS2L)
BIOPAC disposable vinyl electrodes (EL503), 6 electrodes per subject
OPTIONAL: BIOPAC Headphones (OUT1)
BIOPAC electrode gel (GEL1) and abrasive pad (ELPAD)
Computer system
Biopac Student Lab 3.7
Subject Population:
The population to be included in this research is male or female subjects between heights of 5’ and 5’9” with a lean build, and 18-22 years of age. It is imperative that the test subjects do not have any health/medical conditions that inhibit their range of motion, muscle strength, or hinder their joint health in any way be it flexibility, stiffness, strength etc…. If discomfort while operating the device took place, these medical conditions can become injuries. The test subject will be asked if they have any such medical conditions and will confirm this through signed paperwork describing whether they do or do not have any such conditions. The device is intended for Spinal Muscular Atrophy patients who have small arms due to a lack of muscle mass. We want to get test subjects that physically resemble SMA patients such as low muscle mass and lean arm frames.
Privacy and Confidentiality:
Subjects will be made aware how the results will be used through the attached consent form. In the study, the subjects will be given a number and no names will be used. Subjects may be asked to come back for further study through a phone call or e-mail if there were errors with the results obtained. Private information from the subjects will not be posted on the website. Only their designated numbers may be displayed.
Volunteers will be assigned a number when they agree to test for the first time. All data will only be associated with a number. The list of names and corresponding ID number will be maintained by the design group and kept in a secure location. The information will be kept for the duration of the volunteer’s participation in the study. Upon their withdrawal from the study, the information will be destroyed. The design team will not use the volunteer’s name in any way or connect it with their experimental results.
Informed Consent:
Subjects will be required to thoroughly read and sign a consent form and will be orally told what the procedure will be. A pamphlet with a brief description of the device’s function and its purpose (who it will benefit) will be provided. The design group will assist the participant in putting on and taking off the device. The subjects will then be told to raise their arm in order to activate the device to lift upward. Next, they will then be told to lower the device by pressing on the bottom sensor. This will be performed numerous times until enough data is acquired on BIOPAC.
EMG is a technique for evaluating and recording the electrical activity produced by the skeletal muscles. We will use surface electrodes and a computerized data acquisition system to monitor muscle activity (or electromyograms) to test muscle activity when performing the motion without the device and while performing the motion with the device. Surface EMG, a non-invasive test, as opposed to intramuscular EMG using needles and wire, will be used to test the functionality of the device. The adhesive surface electrodes will be placed on the biceps and triceps. The adhesive allows for the easy removal after testing is complete. An electromyogram detects the electrical potential generated by the muscle cells when these cells are neurologically activated. The electrical source is the muscle membrane potential of about -90 mV. EMG potentials range between 50 μV and up to 30 μV. Typical repetition rate of muscle motor firing is about 7-20Hz which depends on the size of the muscle. Muscle tissue at rest is normally electrically inactive. When the muscle is voluntarily contracted, action potentials begin to appear. As the strength of the muscle contraction is increased, more and more muscle fibers produce action potentials. When the muscle is fully contracted, there will be a group of action potentials of varying rates and amplitudes. With the device on, the muscle activity during muscle contraction should be significantly less than without the device.
The patient will be secured into the device by the sensor cuff on the wrist as well as straps around the arm and bicep. The metal frame will support the arm though the patient will not be in contact with the metal. The device will have cushioning material to provide comfort and electrical insulation. Any metal part of the frame that would be in contact with the subject will be insulated.
Each subject will have the same tests performed and it will take about the same amount of time. Test subjects will be studied once during the semester. Testing is not expected to take more than an hour. There is no justified need for follow up trials. Computer software (BIOPAC STUDENT LAB) as well as surface electrodes will be utilized in order to acquire muscle activation data that will be analyzed. This data will consist of an EMG that will be interpreted by the group to determine if the device is working adequately. We plan on testing this device on 20 participants. Each participant will be asked to perform the same motion with and without the device. These will be compared to determine how closely the device approaches physiological range of motion. The study will take place at The College of New Jersey. The investigator s include all team members: Christina Paparella, Andrew Levitsky, Danny Molina, Greg VanNess and Christian Mejia. Christina, Danny and Andrew have performed a similar lab using BIOPAC and specifically testing EMG in the Fall of 2010 in Physiological Systems Laboratory I. The design team has the BIOPAC EMG lesson used in the lab and will follow procedures for interpreting data accordingly.
In order to test the device, BIOPAC data acquisition unit (MP36) will be used to convert the electrical signal of the muscles into interpretable data on a computer. BIOPAC disposable vinyl electrodes (El503) are surface electrodes that are placed on the skin in order to measure motor neuron impulses to be sent to the computer acquisition unit. For optimal electrode adhesion, the electrodes should be placed on the skin at least five minutes before the start of the calibration procedure. Each of the pinch connectors on the end of the electrode cable needs to be attached to a specific electrode. The electrode cables are each a different color to ensure that each cable is connected to the proper electrode. Other important components for testing include the BIOPAC electrode lead set (SS2L), BIOPAC electrode gel (GEL1) and abrasive pad (ELPAD), computer system and BIOPAC Student Lab 3.7.
List of Materials:
BIOPAC data acquisition unit (MP36)
BIOPAC electrode lead set (SS2L)
BIOPAC disposable vinyl electrodes (EL503), 6 electrodes per subject
OPTIONAL: BIOPAC Headphones (OUT1)
BIOPAC electrode gel (GEL1) and abrasive pad (ELPAD)
Computer system
Biopac Student Lab 3.7
Subject Population:
The population to be included in this research is male or female subjects between heights of 5’ and 5’9” with a lean build, and 18-22 years of age. It is imperative that the test subjects do not have any health/medical conditions that inhibit their range of motion, muscle strength, or hinder their joint health in any way be it flexibility, stiffness, strength etc…. If discomfort while operating the device took place, these medical conditions can become injuries. The test subject will be asked if they have any such medical conditions and will confirm this through signed paperwork describing whether they do or do not have any such conditions. The device is intended for Spinal Muscular Atrophy patients who have small arms due to a lack of muscle mass. We want to get test subjects that physically resemble SMA patients such as low muscle mass and lean arm frames.
Privacy and Confidentiality:
Subjects will be made aware how the results will be used through the attached consent form. In the study, the subjects will be given a number and no names will be used. Subjects may be asked to come back for further study through a phone call or e-mail if there were errors with the results obtained. Private information from the subjects will not be posted on the website. Only their designated numbers may be displayed.
Volunteers will be assigned a number when they agree to test for the first time. All data will only be associated with a number. The list of names and corresponding ID number will be maintained by the design group and kept in a secure location. The information will be kept for the duration of the volunteer’s participation in the study. Upon their withdrawal from the study, the information will be destroyed. The design team will not use the volunteer’s name in any way or connect it with their experimental results.
Informed Consent:
Subjects will be required to thoroughly read and sign a consent form and will be orally told what the procedure will be. A pamphlet with a brief description of the device’s function and its purpose (who it will benefit) will be provided. The design group will assist the participant in putting on and taking off the device. The subjects will then be told to raise their arm in order to activate the device to lift upward. Next, they will then be told to lower the device by pressing on the bottom sensor. This will be performed numerous times until enough data is acquired on BIOPAC.